Potato blight tracking in Europe - 2017
EuroBlight is continuously examining the ongoing evolution of the European population of the potato late blight pathogen and now reports on the 2017 results. Almost 1500 samples were genotyped from 16 countries last growing season.
Preliminary conclusions:
- Dominance of some clones across European crops shows EU growers/industry share management challenges
- New clones (EU_36_A2, EU_37_A2 and EU_41_A2) continue to spread in 2017 and are displacing other populations
- Much primary inoculum is locally generated and spread. Better management of inoculum sources would aid management
- Genotype group “others” is highly diverse, ephemeral and most likely the result of sexual oospore germination
- High genetic diversity increases the risk of blight management problems due to a greater probability of pathogen types evolving virulence against novel host resistance genes and reduced sensitivity to specific fungicide active ingredients
- Phenotypic traits of existing and novel genotypes continue to be examined in ERA-NET project (IPMBlight2.0) and by other EU research teams
What is new?
Around 75% of the samples belonged to defined clonal lineages also observed in previous seasons. Some clones are widespread and have been present in Europe for more than a decade, but three emerging clones (EU_37_A2, EU_36_A2 and EU_41_A2) increased their combined frequency from 10% in 2016 to 28% of the population in 2017. Over the same period, genotype 13_A2 decreased from over 30% to less than 20% of the population. A quarter of the population, mainly from the Nordic part of Europe, comprised ephemeral, genetically diverse isolates consistent with oospore-borne inoculum. A clear regional pattern in the dominance of clones versus sexual recombinants was observed across Europe. Some implications of these displacements and ongoing changes are discussed.
How did we do it?
Since its arrival in the nineteenth century, Phytophthora infestans, the cause of potato late blight, has remained a serious threat to European potato production. Although we are now better equipped to control the disease than in the past, evolving pathogen populations continue to challenge integrated management practices. Rapid changes in P. infestans populations causing late blight in Europe, America and Asia, including the emergence of strains with increased aggressiveness or reduced fungicide sensitivity, have been observed. Indeed, the changes in P. infestans populations directly influence the development and deployment of resistant cultivars, the performance of disease warning systems and the efficacy of plant protection products.
Therefore, coordinated and continuous pathogen monitoring was proposed by the EuroBlight consortium at its meeting in 2013, and now implemented as an EU-wide monitoring activity, including all stakeholders. We continue to monitor populations and characterise the invasive genotypes to help optimise IPM strategies, as required by EU Directive 2009/128/EC on the sustainable use of plant protection products.
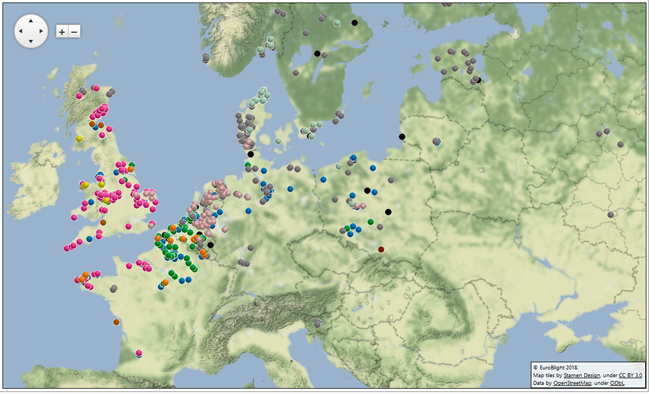
As in previous years, ‘FTA cards’ were distributed to disease ‘scouts’ across the industry, who visited blight-infected crops. Diseased leaves were pressed onto the cards and returned to the laboratories where the pathogen DNA was fingerprinted at the James Hutton Institute and INRA Rennes. The DNA fingerprint data defines the clonal lineages of the pathogen, and is combined with geo-location data to plot the diversity across Europe. Disease pressure in 2017 was lower than that in 2016, which reduced the sampling frequency and distribution slightly.
Around 1500 samples were collected by more than 20 partners across 16 European countries. As in previous years, the AHDB Potatoes ‘Fight Against Blight’ campaign data from crops in Great Britain (GB) is included. Included also is data from partners in the IPMBlight2.0 project, which is also generating pathogen phenotype data to support IPM strategies. The combined data from 2013-2017 now comprise over 6347 samples from 34 countries. With the support of international groups, data was also entered for parts of Asia, South America and North Africa.
What did we discover?
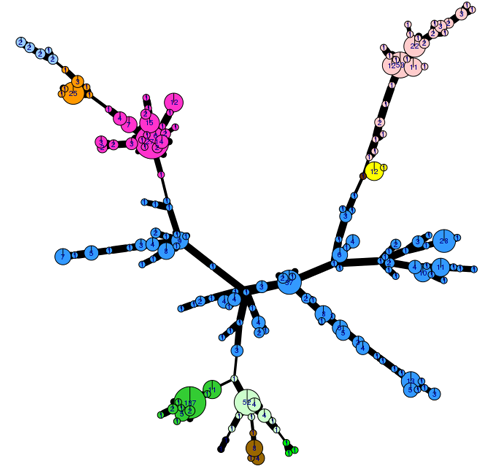
Over the last five years, 60-79% of the population comprised known clonal lineages that recur each season. The remaining samples were novel, genetically diverse genotypes mostly found at a single location in one season and grouped in a category termed ‘Other’ (Figure 1).
 |  |
|---|---|
See graphic in large size here |
In 2017, clone EU_13_A2 (blue-13) declined from 31% of the samples to 19% which, for the first time, was lower than that of EU_6_A1 (23%). The global distribution of the aggressive clone EU_13_A2 and its resistance to metalaxyl continues to affect management efficacy in Europe, parts of Asia and North Africa, reinforcing the need for pathogen data to support IPM best practices. The frequency of EU_6_A1 was stable, but this clone remained localised to GB, France and a single sample from Belgium. The frequency of EU_1_A1 further decreased from 4.5 to 2.2% of the population, and was found in the same countries as EU_6_A1.
Three new clones have been displacing the above genotypes and increased their range in 2017. EU_36_A2 was first sampled at low frequencies in the starch potato areas in Germany and the Netherlands in 2014 and 2015. Over 2016 and 2017, its incidence increased from 3.7 to 8.8% of the population, and it has spread across the Netherlands into Belgium, GB, Denmark and Poland. Genotype EU_37_A2 was first detected in Noordoostpolder in the Netherlands in 2013, and remained local at low frequency in 2014 and 2015. It increased to 5.5% of the population sampled in 2016, and in 2017 reached 14.4%, also becoming established in England, Belgium and northern France. The third emerging genotype is a clone called EU_41_A2, first recorded in Denmark in 2013. It has now become established, making up 4.5% of the 2017 population and having spread to southern Norway and Sweden and to northern Poland in a pattern consistent with airborne dispersal. The result of the mating type test For EU_41 was released on 19 April and it showed clearly to be the A2 mating type. The survival and spread of these clones, when others are decreasing or have failed to establish, suggests they are evolutionarily fit and may pose challenges to disease control. Reduced sensitivity to fluazinam was reported in EU_37_A2 by Wageningen University in 2017, which is having implications on management practices. However, no information is yet available on the sensitivity of the other clones to fungicide active ingredients.
Lastly, the genetically diverse ‘Other’ samples comprised 25% of the population, and remained at the highest frequency in the east and north-east of Europe. In contrast to some clones, the traits possessed by the ‘Other’ isolates and other clones are not yet known. However, the IPMBlight2.0 project (http://euroblight.net/research-projects/ipmblight20) has started a detailed study of the traits of clonal and sexually recombinant isolates to help improve late blight management and forecasting.
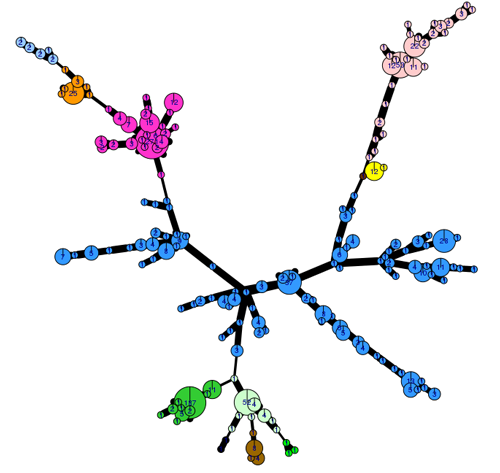
The genetic diversity of the 2017 population can be visualised using an analysis tool (poppr 2.0) linked to the EuroBlight pathogen database. The minimum spanning network (Fig. 2) shows the 2017 population structure, only including identified clones. Considerable sub-clonal diversity within the 13 year old EU_13_A2 lineage is observed compared to that in the ‘younger’ clones. ‘Other’ isolates (not shown) are genetically diverse and distributed across the whole network. Detailed analysis is underway to examine population change using these tools.
 |  |
|---|---|
See graphic in large size here |
The EuroBlight model of pathogen tracking is a rapid, cost-effective and co-ordinated approach to understanding pathogen evolution on a European scale. Data on the dominant clones has been passed to growers, advisors, breeders and agrochemical companies to provide practical management advice and shape longer-term strategies. The data provides an early warning of the incidence and spread of novel clones that is enabling a timely response from the industry.
The EuroBlight network is continuing to harmonise methods with other networks in the Americas and Asia and encourages continued co-operation between groups involved in managing late blight to exploit the database and tools for improved awareness and blight management on a global scale. We will continue the project in 2018, so please make contact with the project team if you would like more information. We thank all the partners who have contributed to the funding and data collection.
Companies and institutions that participated in the sampling and sponsored the project (2013-2017)
Aarhus University, ACVNPT, ADAMA, AFBI, Agrifirm, Agricultural Institute of Slovenia, Agriphar, AHDB Potatoes, ARVALIS-Institut du Végétal, BASF SE, Bayer CropScience AG, Bayerische Landesanstalt für Landwirtschaft, Belchim Crop Protection, Centre Wallon de Recherches Agronomiques, Certis, Cheminova, CropSolutions, CZAV, Delphi, Dupont de Nemours, Emsland Group, Estonian University of Life Sciences, Germicopa SAS, HZPC Holland B.V., INRA, The James Hutton Institute, Neiker, Nordisk Alkali, NIBIO, PCA, The Plant Breeding and Acclimatization Institute (IHAR), Profytodsd, Swedish University of Agricultural Sciences, Syngenta Agro GmbH and Wageningen University and Research.
Contacts
- Jens G. Hansen & Poul Lassen, Aarhus University: Contact: jensg.hansen@agro.au.dk
- Geert Kessel, Wageningen University and Research Centre. Contact: geert.kessel@wur.nl
- David Cooke: James Hutton Institute, Dundee. Contact: david.cooke@hutton.ac.uk
- Didier Andrivon, Roselyne Corbiere: Contact: Didier.Andrivon@.inra.fr
Download the news story as pdf here
How to participate? Sampling protocol and sampling form is updated here
 |  |